Vascular Tumors Result from Adeno-Associated Virus-9 Angiogenic Gene Therapy of Bone Allografts Vascular Tumors
Main Article Content
Background: Cryopreserved bone allografts are often used to reconstruct segmental bone defects. They are non-viable, which can result in infection, non-union and stress-fractures. We aimed to revascularize allografts in porcine and rat models using vascular endothelial growth factor (VEGF), combined with platelet derived growth factor (PDGF) administered through an adeno-associated viral vector. We report the development of vascular tumors resulting from this treatment.
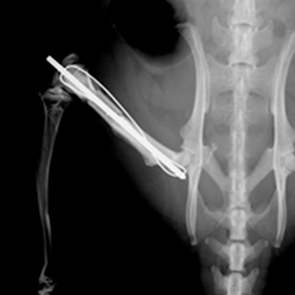
Methods: In two separate studies, an identical AAV.VEGF.PDGF vector was used to promote angiogenesis in cryopreserved bone allografts. In 8 Yucatan minipigs, a 3.5 cm segmental tibial defect was reconstructed with a matched allograft, revascularized by placement of a transfected arteriovenous (AV) bundle within the medullary canal. In another experiment, cryopreserved femoral bone allografts coated with AAV.VEGF.PDGF were placed across a 10 mm segmental femoral gap in 10 Lewis rats.
Results: Vascular tumors developed in skin and subcutaneous tissues in 5 out of 8 pigs and all of the rats. Histology revealed changes essentially identical to those seen in pyogenic granuloma (lobular capillary hemangioma) in humans. Polymerase chain reaction (PCR) identified the sequence of human VEGF-DNA in all of the sampled tumor tissues.
Conclusion: Recombinant AAV gene therapy used to promote angiogenesis in avascular bone risks the development of vascular cutaneous lesions. Gene therapy using an identical AAV.VEGF.PDGF vector should not be considered clinically until safe use can be demonstrated and.concerns regarding chromosomal integration, dose effect and species differences are addressed.