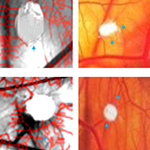
Angiogenic efficacy of Heparin on chick chorioallantoic membrane
Main Article Content
Abstract
Heparin is an anticoagulant agent known to have diverse effects on angiogenesis with some reports suggesting that it can induce angiogenesis while a few have indicated of its inhibitory property. Cancer patients treated for venous thromboembolism with low molecular heparin had a better survival than the unfractionated heparin (UFH). Heparin is known to interact with various angiogenic growth factors based on its sulfation modifications within the glycosaminoglycan chains. Therefore it is important to study the mechanism of action of heparin of different molecular weight to understand its angiogenic property. In this concern, we examined the angiogenic response of higher molecular weight Heparin (15 kDa) of different concentrations using late CAM assay. Growth of blood vessels in terms of their length and size was measured and thickness of the CAM was calculated morphometrically. The observed increase in the thickness of the CAM is suggestive of the formation of capillary like structures at the treated region. Analysis of the diffusion pattern showed internalized action of heparin that could affect gene expression leading to proliferation of endothelial cells. Angiogenesis refers to formation of new blood vessels from the existing ones and occurrence of new blood vessels at the treated area strongly confirms that heparin of 15 kDa molecular weight has the ability to induce angiogenesis on CAM vascular bed in a dose dependent manner. The results demonstrate the affinity of heparin to induce angiogenesis and provide a novel mechanism by which heparin could be used in therapeutics such as in wound healing process.
Article Details
How to Cite
REMA, Reji Bhuvanendran; RAJENDRAN, Karthick; RAGUNATHAN, Malathi.
Angiogenic efficacy of Heparin on chick chorioallantoic membrane.
Vascular Cell, [S.l.], v. 4, n. 1, p. 8, apr. 2012.
ISSN 2045-824X.
Available at: <https://vascularcell.com/index.php/vc/article/view/10.1186-2045-824X-4-8>. Date accessed: 08 june 2026.
doi: http://dx.doi.org/10.1186/2045-824X-4-8.
Section
Original Research